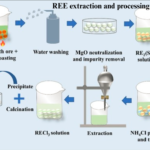
What Are Sulfate Minerals?
Sulfate minerals are a class of minerals that contain the sulfate anion (SO₄²⁻) combined with metals such as calcium, barium, lead, or magnesium. These minerals typically form in evaporitic environments, hydrothermal veins, and as oxidation products of sulfide minerals. They are widely used in various industrial applications, including mining, construction, chemical production, and environmental management.
Importance of Sulfate Minerals in the Mining Industry
1. Ore Sources for Industrial and Metal Extraction
Some sulfate minerals serve as direct sources of valuable metals and industrial materials.
Key Examples:
- Barite (BaSO₄)
- Used extensively in oil and gas drilling as a weighting agent in drilling fluids.
- A critical component in barium-based chemicals, used in paints, medical imaging (barium contrast agents), and glass manufacturing.
- Extracted from both primary sulfate mineral deposits and hydrothermal veins.
- Anglesite (PbSO₄)
- A secondary mineral derived from the oxidation of galena (PbS) in lead ore deposits.
- Mined as a significant source of lead for the production of batteries, radiation shielding, and alloys.
- Alunite (KAl₃(SO₄)₂(OH)₆)
- Previously mined as an aluminum ore, particularly during wartime shortages of bauxite.
- Contains potassium sulfate, which is used in fertilizers.
2. Role in Mining and Environmental Applications
Sulfate minerals play a crucial role in mine site management and environmental sustainability.
- Gypsum (CaSO₄·2H₂O) & Anhydrite (CaSO₄)
- Used for mine land reclamation by improving soil quality and neutralizing acidic mine tailings.
- Essential in cement manufacturing, reducing environmental waste by utilizing mining byproducts.
- Epsomite (MgSO₄·7H₂O) & Melanterite (FeSO₄·7H₂O)
- Indicators of mine drainage oxidation and the presence of sulfide minerals.
- Used in water treatment to remediate acid mine drainage (AMD) by neutralizing acidity.
3. Byproducts of Sulfide Ore Processing
Many sulfate minerals form as byproducts of mining operations, particularly in the oxidation zones of sulfide deposits.
- Jarosite (KFe₃(SO₄)₂(OH)₆)
- Commonly found in acid mine drainage and tailing ponds.
- Used in environmental studies to monitor the impact of mining waste.
- Schwertmannite (Fe₈O₈(OH)₆(SO₄)₂)
- Forms in iron-rich acidic waters at abandoned mines.
- Acts as a natural sorbent for heavy metals, helping in pollution control.
4. Contribution to Sulfuric Acid Production
Sulfate minerals contribute to the production of sulfuric acid (H₂SO₄), a key chemical in mining and metallurgy.
- Pyrite (FeS₂) & Other Sulfide Minerals → Sulfuric Acid
- Sulfuric acid is crucial in ore leaching processes, especially in the extraction of copper, uranium, and nickel.
- Used in acid leaching to process low-grade ores in heap leaching operations.
- Gypsum as a Byproduct of Sulfuric Acid Neutralization
- During the production of sulfuric acid, gypsum (CaSO₄·2H₂O) forms as a byproduct, which can be repurposed in construction and agriculture.