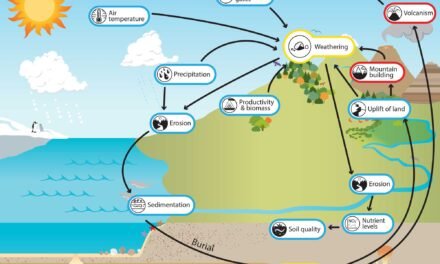
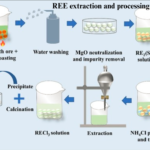
Hydroxide mineral deposits form under specific geological and environmental conditions, typically through weathering, oxidation, and hydrothermal processes. The formation of these deposits often involves the interaction of metal cations with water, leading to the precipitation of hydroxide ions (OH⁻) and the subsequent formation of minerals.
Formation of Hydroxide Mineral Deposits
- Weathering of Primary Minerals:
- Hydroxide minerals often form as a result of the weathering of primary minerals (like feldspar, olivine, and pyroxenes) in the Earth’s crust. When these minerals are exposed to rainwater, oxygen, and carbon dioxide, they break down, and their metal cations (such as iron, aluminum, and magnesium) react with water to form hydroxide minerals.
- For example, gibbsite (Al(OH)₃) forms when aluminum-rich minerals, like feldspar, weather and release aluminum ions that then combine with water to form gibbsite.
- Oxidation of Metal Sulfides:
- Oxidation of metal sulfides, particularly those containing iron (such as pyrite), can result in the formation of hydroxide minerals like goethite (FeO(OH)) and limonite (FeO(OH)·nH₂O). This occurs when oxygen and water interact with metal sulfides in ore deposits, leading to the formation of iron hydroxides.
- The oxidation of iron in sulfide ores like pyrite often occurs near the Earth’s surface, where acid mine drainage (AMD) may also occur, causing the mineralization of iron hydroxides.
- Hydrothermal Processes:
- In hydrothermal environments (i.e., areas where hot water or steam circulates through the Earth’s crust), certain hydroxide minerals can form as secondary minerals. This occurs when hot water leaches metal ions from surrounding rocks, which then precipitate as hydroxide minerals in cooler areas of the hydrothermal system.
- Brucite (Mg(OH)₂), for example, forms from the interaction of magnesium-rich rocks with water under specific temperature and pressure conditions.
- Evaporation and Precipitation in Alkaline Lakes:
- In certain alkaline or saline lakes, the evaporation of water can cause the precipitation of hydroxide minerals. This is often seen in environments with high pH where minerals such as hydrozincite (Zn₅(CO₃)₂(OH)₆) form as a result of evaporation and the concentration of metal ions in solution.
- Biological Influence:
- Microbial activity can also contribute to the formation of hydroxide minerals, particularly in environments where microorganisms play a role in the oxidation of metal ions. Some microbes can oxidize iron, promoting the formation of iron hydroxides like goethite and limonite.
- Serpentine minerals can form in environments where biological weathering of ultramafic rocks (rich in magnesium) occurs, possibly influenced by microbial and biogeochemical processes.
Largest Global Sources of Hydroxide Minerals
- Gibbsite (Aluminum Hydroxide):
- Bauxite Deposits: The largest source of gibbsite is bauxite, which is the primary ore for aluminum production. Bauxite forms from the weathering of aluminum-rich rocks in tropical and subtropical climates.
- Major Global Sources:
- Australia: The world’s largest producer of bauxite, with massive deposits in Western Australia (such as the Weipa and Gove deposits).
- Guinea: One of the leading sources of bauxite, particularly from the Fria and Boké regions.
- Brazil: Significant bauxite deposits are found in Amazônia and Minas Gerais.
- India: Bauxite is found in significant amounts in Orissa, Andhra Pradesh, and Gujarat.
- Goethite (Iron Hydroxide):
- Iron Ore Deposits: Goethite is a common iron ore and is often found as part of limonite deposits. These ores are typically oxidized and weathered iron-bearing minerals.
- Major Global Sources:
- Australia: Goethite is a significant component of Australia’s iron ore deposits, especially in the Pilbara region.
- Brazil: The Carajás mineral province is rich in iron ore, including goethite-rich deposits.
- China: Known for significant iron ore mining, including iron hydroxides.
- India: Goethite is found in several regions, particularly in the Odisha and Jharkhand states.
- Limonite (Iron Hydroxide):
- Iron Ore Deposits: Limonite, a mixed iron oxide-hydroxide mineral, is often found in the weathered zones of iron ore deposits. It is less common than goethite but still an important source of iron.
- Major Global Sources:
- West Africa: Countries like Liberia, Sierra Leone, and Guinea contain notable limonite deposits.
- United States: The Great Lakes region contains some limonite-rich iron ore deposits, particularly in Michigan and Minnesota.
- Serpentine (Magnesium Hydroxide):
- Magnesium-rich Rocks: Serpentine forms from the alteration of ultramafic rocks, particularly in serpentinized peridotites and dunites.
- Major Global Sources:
- China: One of the largest producers of serpentine minerals, particularly in Xinjiang and Jilin regions.
- United States: The Pacific Coast and areas in California contain large serpentine deposits.
- Russia: Serpentine is abundant in regions like Eastern Siberia and the Urals.
- Brazil: Large deposits of serpentine minerals are found in Minas Gerais and Amapá.
- Hydrozincite (Zinc Hydroxide Carbonate):
- Zinc Ore Deposits: Hydrozincite forms as a secondary mineral from the oxidation of primary zinc ores.
- Major Global Sources:
- Australia: Known for large zinc deposits that can contain hydrozincite, particularly in Queensland and New South Wales.
- China: Significant zinc mining operations that include secondary minerals like hydrozincite.
- United States: The Mississippi Valley-type deposits in Missouri and Tennessee include hydrozincite as a secondary mineral.
- Brucite (Magnesium Hydroxide):
- Magnesium-rich Rocks: Brucite forms in alkaline environments or through hydrothermal processes from magnesium-rich rocks.
- Major Global Sources:
- China: One of the largest producers of brucite, especially in Xinjiang and Inner Mongolia.
- United States: Brucite deposits can be found in California and Michigan.
- Russia: The Ural Mountains contain significant amounts of brucite
Hydroxide mineral deposits form through a variety of geological processes, including weathering, oxidation, hydrothermal activity, and biological processes. They are often found in tropical and subtropical environments, as well as in areas with acidic or alkaline conditions.