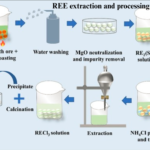
The extraction of hydroxide minerals like bauxite (aluminum hydroxide) and goethite (iron hydroxide) involves several key methods that depend on the specific mineral and its form within the ore. These methods primarily include surface mining, hydrometallurgical processes, and pyrometallurgical techniques.
Here’s a detailed overview of the primary extraction methods for these hydroxide minerals:
1. Extraction of Bauxite (Aluminum Hydroxide)
Bauxite is the main source of aluminum and consists primarily of aluminum hydroxide minerals like gibbsite, boehmite, and diaspore. The primary method for extracting aluminum from bauxite is the Bayer Process, which involves refining bauxite into alumina (Al₂O₃), a precursor to aluminum metal.
Primary Extraction Methods:
- Open-Pit Mining:
- Bauxite is typically found in shallow deposits, so open-pit mining is the most common method for extraction. This method involves removing the overburden (soil and rock) to expose the bauxite ore beneath.
- Excavators, draglines, and haul trucks are used to extract the bauxite, which is then transported to processing plants.
- Bayer Process (Refining):
- After the bauxite is extracted, it is crushed and ground to a fine powder.
- The Bayer process involves dissolving the bauxite in a high-temperature sodium hydroxide (NaOH) solution at temperatures of about 150–200°C under pressure. This forms a solution of sodium aluminate and leaves behind impurities like iron oxides, silica, and other minerals.
- The solution is then allowed to cool, and the aluminum hydroxide precipitates out as gibbsite or boehmite. This is followed by filtration and washing to remove any remaining impurities.
- The gibbsite is then calcined (heated to high temperatures, around 1000–1100°C) in a rotary kiln to remove water and produce alumina (Al₂O₃), which is the final product used to make aluminum metal.
- Alternative Methods:
- Alkaline Leaching: In certain cases, bauxite may undergo an alternative leaching process using different solvents or seawater to extract aluminum compounds.
2. Extraction of Goethite (Iron Hydroxide)
Goethite is an important iron ore that forms through the weathering of other iron-rich minerals, such as magnetite or hematite, in the presence of water and oxygen. Goethite is typically found in oxidized iron ore deposits and is usually mined along with other iron minerals.
Primary Extraction Methods:
- Open-Pit Mining:
- Goethite-rich iron ore deposits are typically extracted using open-pit mining methods, which are similar to those used for bauxite. The overburden is removed to expose the ore, and excavators and haul trucks are used to transport the ore to the processing plant.
- Beneficiation:
- After extraction, the ore is crushed and ground to separate the valuable goethite-rich iron ores from waste material (gangue).
- The goal of beneficiation is to concentrate the iron content while removing as much of the silica, alumina, and other impurities as possible. The beneficiation methods include:
- Gravity Separation: Using differences in density to separate goethite from lighter gangue minerals.
- Magnetic Separation: This can be used if the ore contains magnetite or hematite along with goethite, though goethite itself is not magnetic.
- Flotation: Sometimes used to separate iron minerals from unwanted silicates.
- Direct Reduction (DIDR):
- Direct Reduction of goethite can be done by heating the goethite ore in a shaft furnace with a reducing gas, such as natural gas or hydrogen, to produce direct-reduced iron (DRI).
- Iron ore pellets or lumps of goethite are subjected to high temperatures in a reduction furnace where carbon monoxide (CO) reduces the iron hydroxide (goethite) to iron metal.
- This method is used to produce high-quality iron that can be used directly in steelmaking.
- Blast Furnace Process:
- In many cases, goethite is blended with other iron ores (such as hematite or magnetite) and processed in a blast furnace.
- Coke, limestone, and hot air are introduced into the blast furnace, where the iron ore is reduced to molten iron and slag is removed. The slag can also be recycled in other industrial applications.
- The goethite undergoes a reduction to form iron metal, and the by-product is slag, which is composed of impurities like silica, alumina, and other minerals.
- Hydrometallurgical Process:
- In some cases, goethite may be processed using hydrometallurgical techniques that involve dissolving the iron ore in acids or alkaline solutions. However, this is less common than traditional pyrometallurgical processes like the blast furnace method.
- Leaching with sulfuric acid or ammonium salts can be used to extract iron and separate it from other minerals.
The extraction of hydroxide minerals like bauxite (aluminum hydroxide) and goethite (iron hydroxide) involves well-established mining and processing techniques.