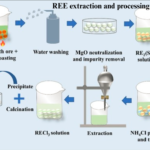
Sulfate minerals play a significant role in metal ore mining and processing, influencing ore extraction, environmental management, and metallurgical processes. Their presence can be both beneficial and challenging, depending on the specific mining environment. Below are the key ways sulfate minerals impact mining operations.
1. Formation of Secondary Metal Ores (Beneficial Impact)
Sulfate minerals can form in the oxidation zones of metal sulfide deposits, leading to the enrichment of valuable metals.
Supergene Enrichment:
- Sulfate minerals such as anglesite (PbSO₄) and jarosite (KFe₃(SO₄)₂(OH)₆) form through the oxidation of sulfide minerals (e.g., galena PbS, pyrite FeS₂).
- These secondary minerals enhance the grade of metal ores by concentrating valuable metals near the surface.
- Supergene enrichment improves the economic viability of ore deposits by making them easier to extract.
Example Deposits:
- Broken Hill, Australia – Anglesite contributes to lead ore formation.
- Rio Tinto, Spain – Jarosite indicates iron-rich supergene mineralization.
2. Acid Mine Drainage (AMD) and Environmental Challenges
One of the most significant negative impacts of sulfate minerals is their role in acid mine drainage (AMD), a major environmental concern in metal mining.
How It Happens:
- Pyrite (FeS₂) and other sulfide minerals oxidize in the presence of water and oxygen, producing sulfuric acid (H₂SO₄).
- The acid dissolves toxic heavy metals (lead, arsenic, cadmium, etc.), contaminating groundwater and surface water.
- Sulfate minerals like jarosite and melanterite (FeSO₄·7H₂O) indicate active AMD formation.
Effects on Mining Operations:
- Increased water treatment costs due to acid pollution.
- Corrosion of mining equipment and infrastructure from acidic conditions.
- Regulatory compliance issues for mining companies.
Mitigation Strategies:
- Lime neutralization – Adding limestone (CaCO₃) to neutralize acidic waters.
- Bioremediation – Using sulfate-reducing bacteria to precipitate heavy metals.
- Mine tailing covers – Preventing oxygen exposure to sulfide-rich waste dumps.
Example Sites with AMD Issues:
- Berkeley Pit, Montana (USA) – A major AMD site with high sulfate and heavy metal concentrations.
- Witwatersrand Gold Mines, South Africa – Long-term AMD issues affecting groundwater.
3. Sulfate Scaling in Metallurgical Processing
During the processing of metal ores, sulfate minerals can cause scaling and fouling in industrial equipment.
How Sulfate Scaling Occurs:
- High sulfate concentrations in process water lead to the precipitation of insoluble sulfates such as gypsum (CaSO₄·2H₂O), barite (BaSO₄), and anhydrite (CaSO₄).
- These minerals deposit inside pipes, heat exchangers, and leaching tanks, reducing operational efficiency.
- Barite scaling is especially problematic in hydrometallurgical plants where barium-rich ores are processed.
Methods to Control Sulfate Scaling:
- Chemical additives (scale inhibitors) – Prevent sulfate precipitation.
- Water treatment & filtration – Removing excess sulfates before processing.
- Alternative leaching solutions – Using chloride-based leaching to avoid sulfate issues.
4. Role in Heap Leaching and Solution Mining
Sulfate minerals influence heap leaching, a technique used to extract metals like copper, uranium, and gold.
Positive Role:
- Gypsum (CaSO₄·2H₂O) in heap leaching:
- Gypsum acts as a permeability enhancer, preventing fine particles from clogging the heap.
- It stabilizes pH levels, improving metal recovery rates.
Negative Role:
- Formation of sulfate crusts can block solution flow, reducing metal extraction efficiency.
- Jarosite precipitation in gold cyanidation interferes with gold recovery, leading to losses.
Example Sites Using Heap Leaching:
- Escondida Copper Mine, Chile – Gypsum controls solution chemistry in leaching piles.
- Nevada Gold Mines, USA – Heap leaching with sulfate minerals influencing recovery.
5. Influence on Tailings Management
Sulfate minerals play a role in the stability and environmental impact of mine tailings.
Challenges:
- High sulfate concentrations in tailings ponds can lead to gypsum precipitation, reducing water recycling efficiency.
- Oxidation of jarosite and other iron sulfates can cause long-term acid drainage problems.
Solutions:
- Covering tailings with impermeable layers to prevent sulfate oxidation.
- Using sulfate-reducing bacteria to convert sulfates into non-reactive sulfides.
Sulfate minerals have both positive and negative effects on metal ore mining and processing. They contribute to supergene enrichment, improving metal recovery, but also cause acid mine drainage (AMD), sulfate scaling in processing plants, and leaching inefficiencies.