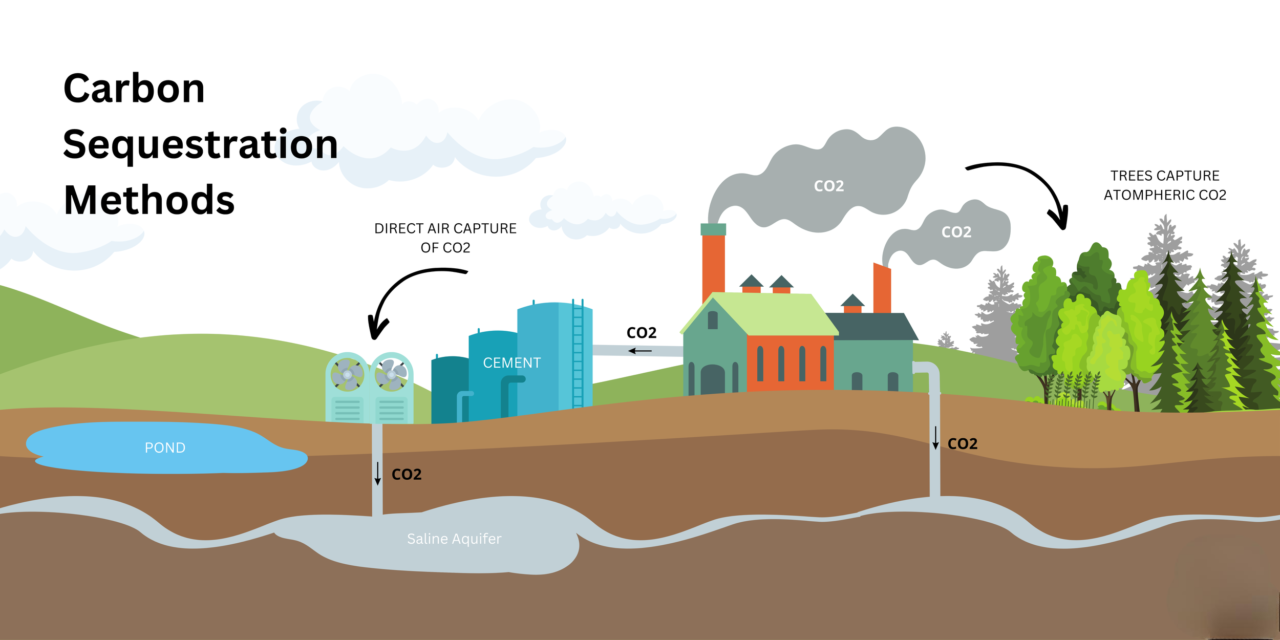
1. CO₂ Sequestration Using Carbonate Minerals
Natural Carbonation : Carbonate minerals like calcite (CaCO₃) and magnesite (MgCO₃) naturally react with CO₂ to form stable carbonates, permanently storing carbon.Mineral Carbonation Process :
Reaction : CO₂ + Ca/Mg-rich minerals → Stable carbonates (CaCO₃ / MgCO₃).Occurs in ultramafic rock deposits containing olivine, serpentine, and brucite .
Enhanced Carbon Sequestration :
Industrial applications : Using mine tailings from nickel, platinum, and asbestos mines to capture CO₂.Accelerated weathering : Injecting CO₂ into carbonate-rich mine waste to speed up sequestration.
2. Carbonate Minerals in Sustainable Mining
Neutralizing Acid Mine Drainage (AMD) :
Limestone (CaCO₃) is used to neutralize acidic waters from sulfide mining, reducing environmental damage.
Reducing Carbon Footprint :
Carbonate-rich tailings can be repurposed for CO₂ capture, making mining operations more sustainable.
Eco-Friendly Construction Materials :
Carbon-negative concrete made using CO₂-reacted minerals, reducing emissions from cement production.
3. Practical Applications in Green Mining
Mine Waste Utilization :
Using mine tailings for CO₂ sequestration instead of disposal.
Carbon-Neutral Mining :
Integrating carbonation technologies in mining to offset CO₂ emissions.
Land Reclamation :
Restoring mined land using carbonate-rich materials to improve soil health and prevent erosion.